Staying Fit
For all the difficult news about the coronavirus in the last nine months — the global death toll of more than 1 million people, failed drug trials, survivors’ lingering symptoms — new drug and other therapies for COVID-19 are starting to emerge. Even as the world waits for a vaccine, researchers are confident they are making progress on treatments that can increase survival rates.
"We know so much more now than we did in March or April,” says Gopi Patel, M.D., associate professor of infectious diseases in the department of medicine at the Mount Sinai Hospital in New York City.


AARP Membership— $12 for your first year when you sign up for Automatic Renewal
Get instant access to members-only products and hundreds of discounts, a free second membership, and a subscription to AARP the Magazine.
For one thing, she says, as hospital caseloads exploded early in the pandemic, patients with milder symptoms were asked to stay at home unless their condition worsened. When patients got sicker and returned to the hospital, they needed higher levels of care, sometimes requiring a ventilator. And once patients are on a ventilator, “the mortality rate is more than staggering,” Patel notes.
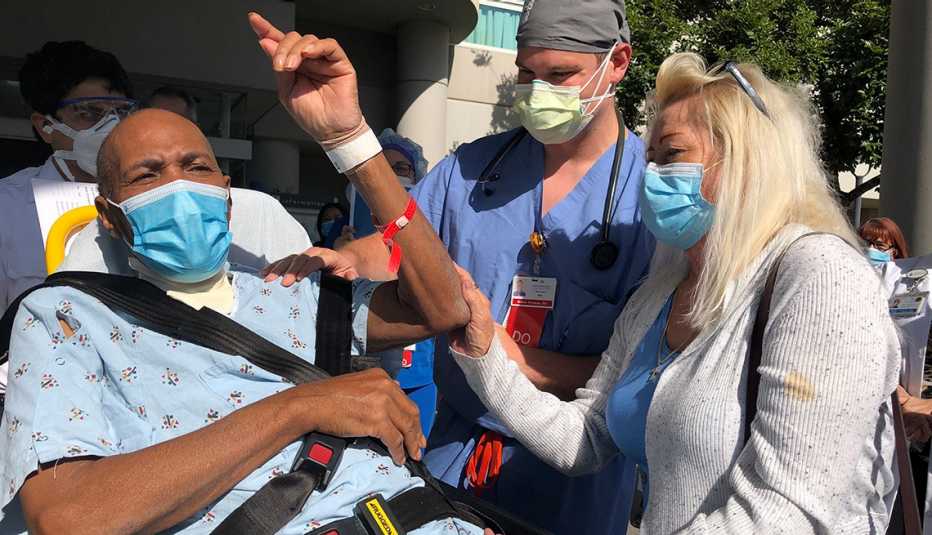
Now, more COVID-19 patients are sent to the hospital for treatment before they get seriously ill, saving lives and resources. In other cases, new therapies can help keep a subset of patients out of the hospital entirely. Here's what doctors say is making a difference now, and what may do so in the future.
Remdesivir and other antivirals
The antiviral remdesivir was approved by the U.S. Food and Drug Administration this week as the first treatment to fight COVID-19, after being shown in clinical trials to help hospitalized patients recover faster from the disease. The drug had previously been used — for months — under an emergency-use authorization. Doctors such as John Beigel, M.D., associate director for clinical research in the division of microbiology and infectious diseases at the National Institute of Allergy and Infectious Diseases, called it “the one antiviral we know that works for SARS-CoV-2 [the coronavirus that causes COVID-19].”
Remdesivir (brand name Veklury), which is given by injection, is now approved for use only in a hospital or a health care setting “capable of providing acute care comparable to inpatient hospital care,” according to the FDA. In announcing their approval, FDA Commissioner Stephen M. Hahn, M.D., stressed that it was supported by safety data from multiple clinical trials and that it represented “an important scientific milestone in the COVID-19 pandemic.” Clinical trials assessing the safety and efficacy of the drug for smaller children are still ongoing, and the drug so far is approved only for those over 12.
A small study published in the New England Journal of Medicine in June showed that when the drug was administered over a 10-day period, 68 percent of hospitalized patients in the study showed clinical improvement. Research also shows that remdesivir works by stopping the virus from spreading in the body.