AARP Hearing Center

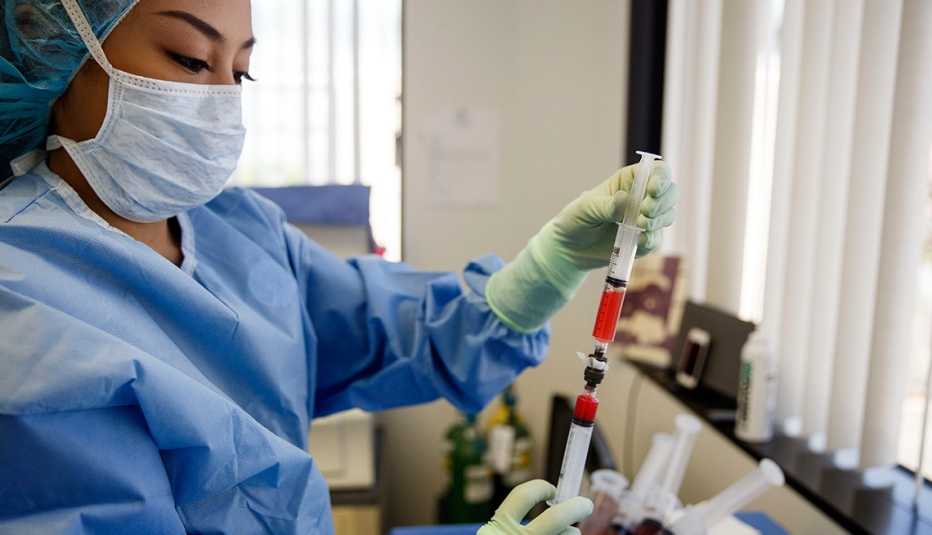
Sitting on the fringes of the mainstream health industry, nearly 600 clinics offering stem cell treatments have sprouted up in the United States. They promise relief from an array of ailments, from spinal cord injury, stroke and Parkinson’s disease to ALS and Alzheimer’s. Stem cell clinics tend to market directly to consumers; their procedures typically are not covered by insurance, and they use a regulatory loophole to perform injection therapies for uses that are not yet approved by the Food and Drug Administration.
The clinics and many satisfied patients herald stem cell treatments as the next great advance in medicine. But critics contend that the current marketplace is based on unproven science. And safety violations at some clinics, which have led to such tragic outcomes as blindness, unusual bone growths and two deaths, have the FDA pondering whether it needs to intervene.
“People have benefited, but the placebo effect can be powerful, especially when you’ve forked over big bucks for the procedure,” says Leigh Turner, a bioethicist at the University of Minnesota and coauthor of a study on the number of domestic stem cell outfits. “And these clinics don’t disclose when patients don’t see any improvement or had serious complications.”
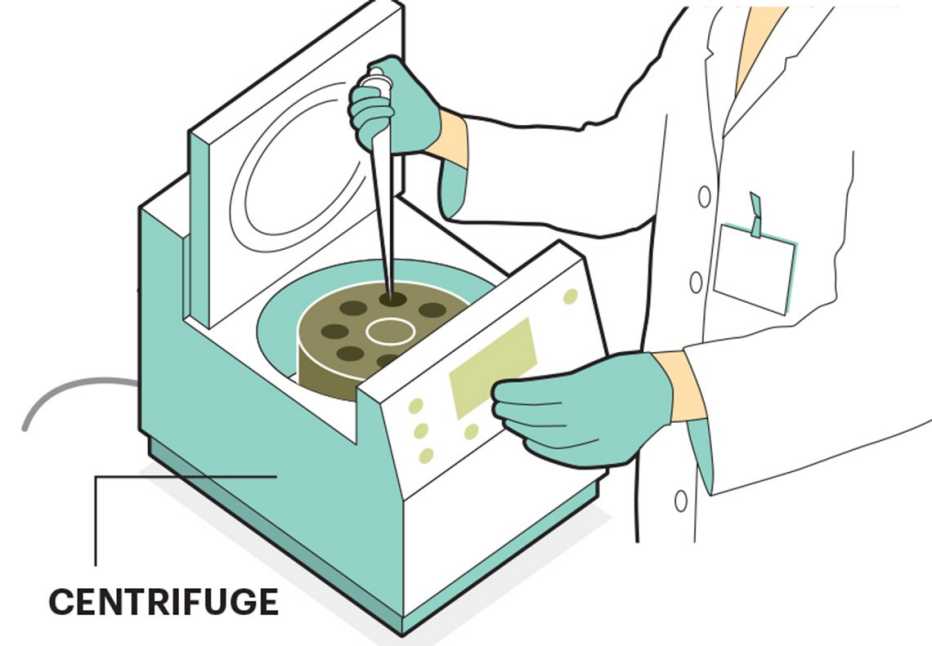
Researchers at academic institutions continue to study the benefits of stem cells, and many see the potential for medical breakthroughs. So far, though, the FDA has approved stem cells only for these treatments: cancer, blood and immune disorders, and skin grafts for burns. But stem cell therapies costing $5,000 to $20,000 per treatment are offered for all sorts of ailments, including those outside the FDA’s approved list, because the clinics operate in a gray area of the law. As long as the patient’s own stem cells are used, and these cells are only subjected to “minimal manipulation,” they’re exempt from federal oversight. That’s because their usage is considered a medical procedure, not a drug.
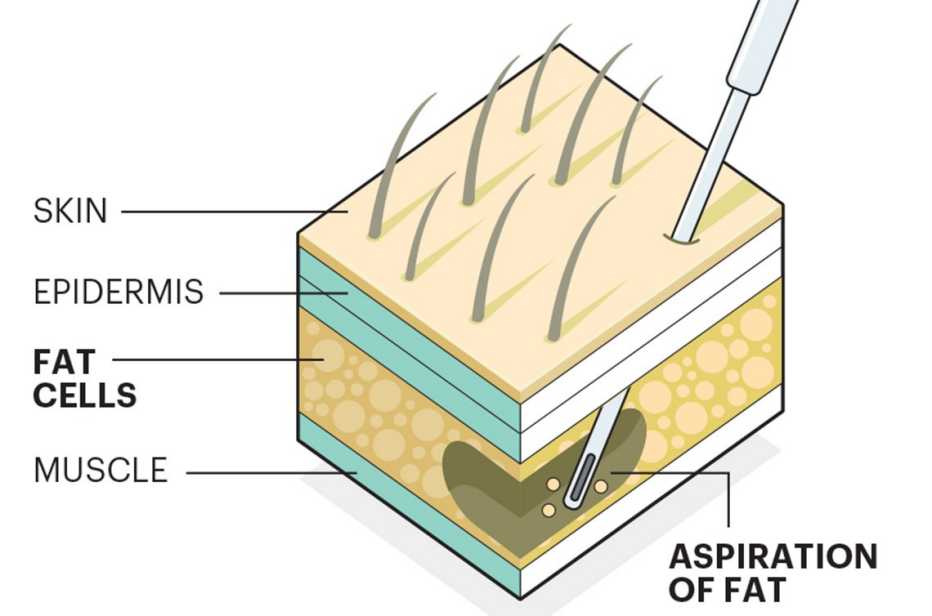
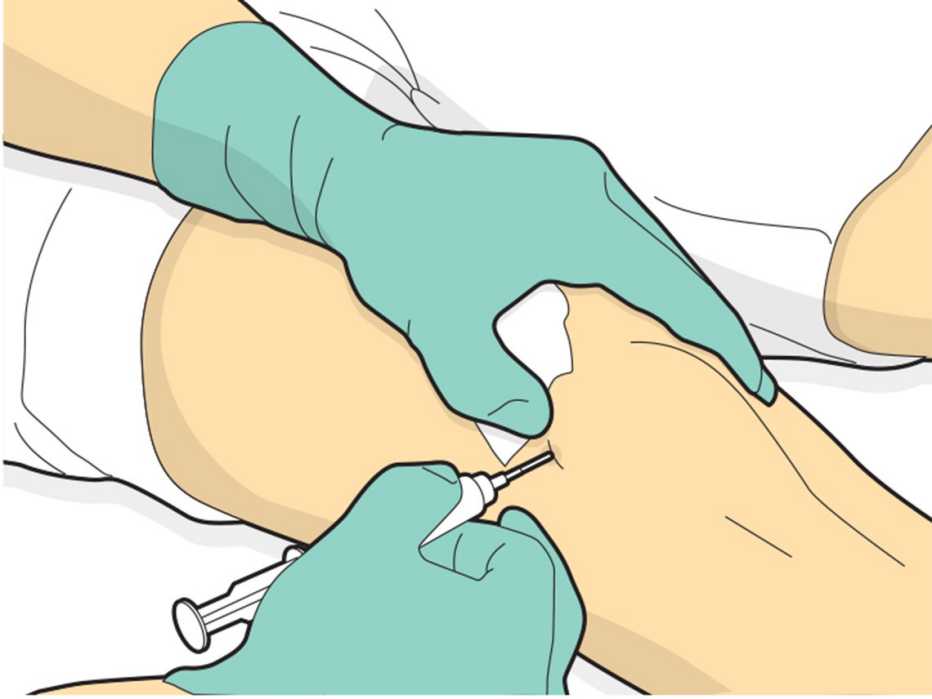
Stem cells are also trendy in cosmetic procedures. For this form of a face-lift, stem cells are injected along with fat to plump up the skin. There’s scant evidence that the procedure works better than standard face-lifts.
What are stem cells?
Stem cells are naturally occurring cells in the body that can develop into nearly any other type of cell. Studies suggest that they work their magic by secreting growth factors (hormonal steroids or proteins) that nurse ailing cells, preventing them from dying. Stem cells also stimulate the formation of blood vessels that nourish damaged tissue with nutrients and oxygen. Stem cells may even form new cells, which could potentially augment the body’s natural repair mechanisms by replacing damaged or dead cells throughout the body.
Twenty years ago, stem cells were touted as the next frontier in medicine. The hope was that we could stick them into an ailing brain or arthritic knee and these grafts would restore lost function as new cells would grow to replace the damaged ones.
The past couple of years have brought notable successes in controlled human experiments, using stem cells developed in labs for more complex disorders such as multiple sclerosis, spinal cord injury, stroke and some forms of blindness. But this research has proceeded rather slowly, meaning it could be a decade or more before such procedures become a standard part of our therapeutic arsenal.










































































More on health
Thyroid Cancer Is the Fastest-Growing Cancer in America
Rates are higher in women than men