Staying Fit
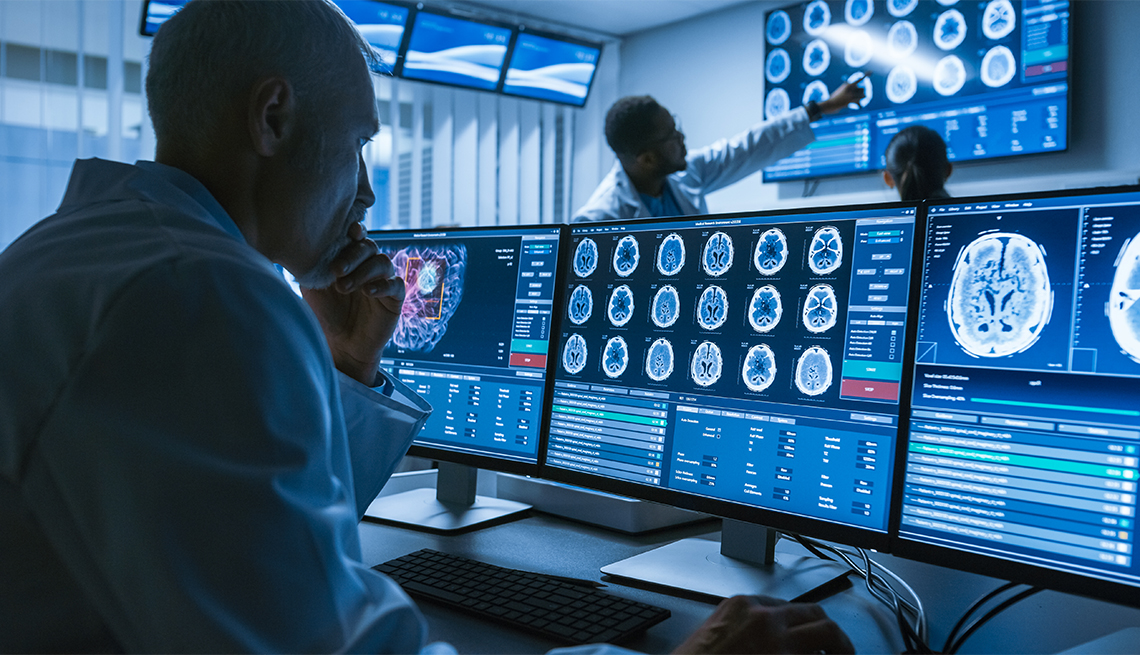
Alzheimer's researchers are exploring a number of new approaches and testing a variety of potential treatments to help the almost 6 million Americans living with the dementia-causing disease.
At a recent gathering — the 12th annual Clinical Trials on Alzheimer's Disease (CTAD) — experts shared some fresh findings, including a new use for a drug now used for Parkinson's disease and new hope from an old drug trial.


AARP Membership— $12 for your first year when you sign up for Automatic Renewal
Get instant access to members-only products and hundreds of discounts, a free second membership, and a subscription to AARP the Magazine.
Promising relief for dementia-related delusions
Hallucinations, delusions and paranoia are common symptoms of Alzheimer's disease and dementia. But for the more than 2 million Americans who experience them, there is no federally approved treatment to curb them.
That could soon change. A drug previously approved for Parkinson's-related psychosis, called pimavanserin, may also help people with dementia, according to results presented at CTAD. Research on the drug was stopped before the trial's end when early results showed the medication could prolong the time between psychotic episode relapses.
The findings are “a big deal,” says Laurie Ryan, an Alzheimer's expert in the Division of Neuroscience at the National Institute on Aging (NIA). Dementia-related psychosis is very difficult for both people with dementia and their caregivers, and it's oftentimes the reason why someone is no longer able to live at home, she explains.
"Some facilities won't even handle patients who have those active symptoms and will send them to an [emergency room], which is really the last thing you want to do for dementia patients because they need the structure” and familiar environments, Ryan adds.
Acadia Pharmaceuticals, which makes pimavanserin, plans to discuss the study's results with the Food and Drug Administration (FDA) in the first half of 2020. If approved for use in people with dementia, pimavanserin would be the first new medicine made available for people living with Alzheimer's in decades.
Potential treatment heads to FDA for approval
Meanwhile, drugmaker Biogen Inc. plans to seek FDA approval in early 2020 for a medicine that could slow mental decline in people living with Alzheimer's disease.