AARP Hearing Center

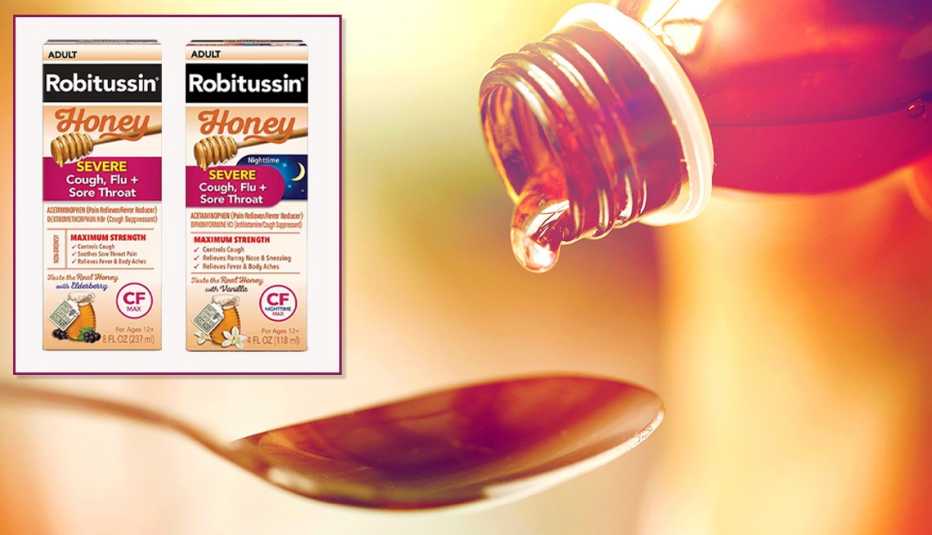
Certain bottles of Robitussin cough syrup have been recalled due to a microbial contamination that may be deadly for people with weakened immune systems, a statement posted by the Food and Drug Administration announced Wednesday.
The manufacturer, Haleon, recalled eight lots of day and nighttime cough syrups used to temporarily relieve cold and flu symptoms, hay fever or other respiratory allergies.
The recalled products pose a risk of “severe or life-threatening events” for individuals with weakened immune systems, including fungemia (presence of fungi or yeast in the blood) or disseminated fungal infection (spread of fungus through the body), the announcement says.

Although life-threatening infections are uncommon for other consumers, there is still a chance of infections that may require medical attention, it says.
There have not been reports of illness related to this recall.
The recalled cough syrup
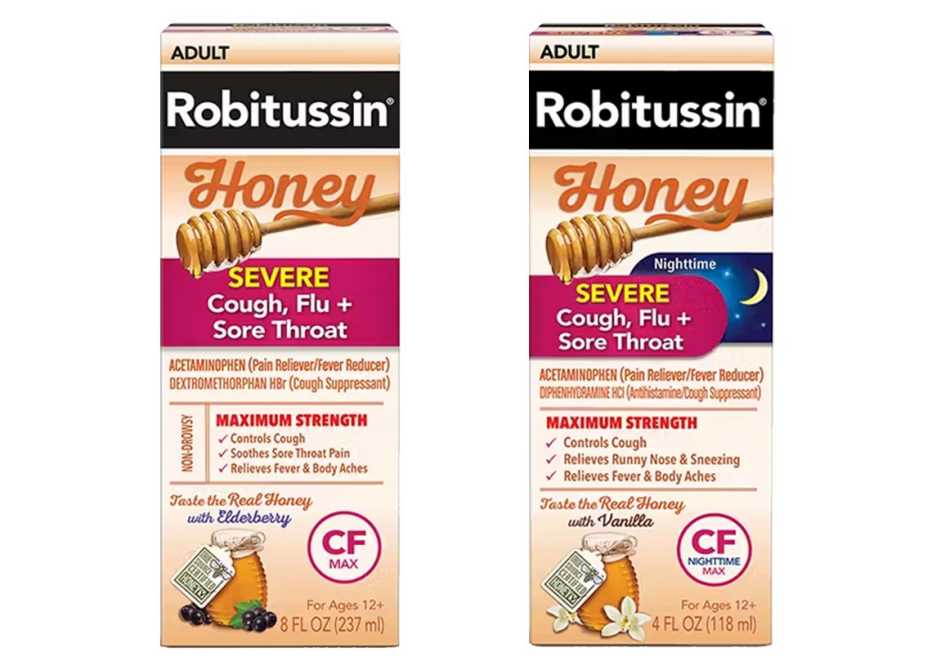
Several varieties of Robitussin Honey CF Max Day and Nighttime were recalled:
- Robitussin Honey CF Max Day Adult sold in 4-ounce bottles, with lot number T10810 and expiring Oct. 31, 2025.
- Robitussin Honey CF Max Day Adult sold in 8-ounce bottles, with lot numbers T08730, T08731, T08732, T08733 and T10808. The affected bottles expire May 31 and Sept 30, 2025.
- Robitussin Honey CF Max NT Adult sold in 8-ounce bottles, with lot numbers T08740 and T08742, expiring June 30, 2026.
Anyone with these products should stop using them and reach out to a health care provider in the event of any negative reactions.
Haleon is notifying distributors and consumers with instructions on how to return the affected products. Questions can be sent to Haleon’s Consumer Relations team at 800-245-1040 or by email to mystory.us@haleon.com.








































































More From AARP
5 Ways to Recover Faster From a Cold
There is no cure-all, but these tips can help relieve exhausting symptoms like stuffiness and coughingMyth-Busting Old Wives’ Tales
When it comes to cold and flu remedies, here’s what works — and what doesn’t
Do You Have a Cold? The Flu? COVID? Or RSV?
A few tricks for telling the illnesses apart
Recommended for You