Staying Fit
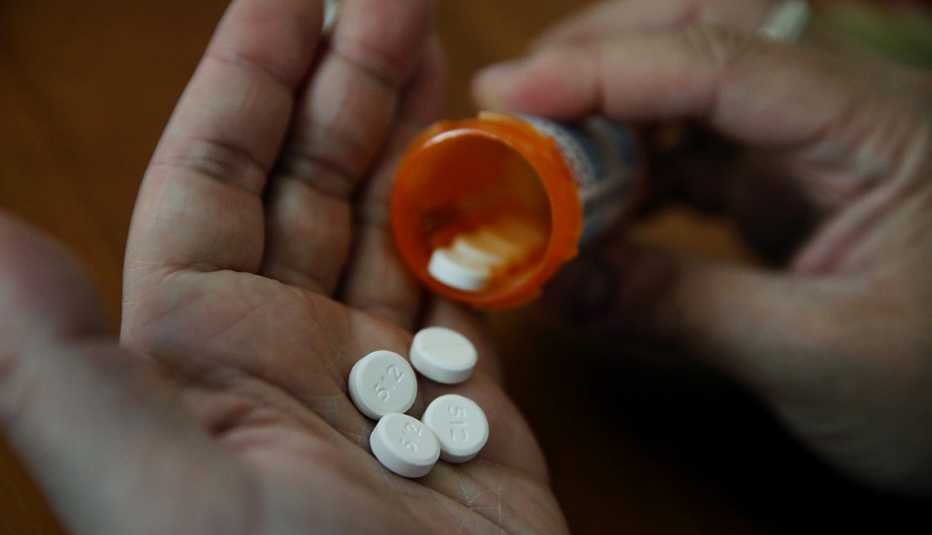
Lupin Pharmaceuticals has recalled four lots of the blood pressure medication Quinapril after detection of the cancer-causing impurity nitrosamine above the acceptable daily intake level set by the Food and Drug Administration (FDA).
Elevated levels of the nitrosamine impurity N-Nitroso-Quinapril were detected in 20 mg and 40 mg tablets of the drug that were shipped nationwide. The recalled lot numbers are G102929, G100533, G100534, G203071 with expiration dates including April 2023, December 2022 and March 2024. See the FDA’s announcement for additional details.


AARP Membership— $12 for your first year when you sign up for Automatic Renewal
Get instant access to members-only products and hundreds of discounts, a free second membership, and a subscription to AARP the Magazine.
Advice to consumers
Patients taking the recalled tablets are advised to continue taking the medication and contact their pharmacist, physician or medical provider to discuss alternative treatment.
Consumers with questions regarding this recall should contact Inmar Rx Solutions Inc. at 877-538-8445 between 9 a.m. and 5 p.m. EST Monday through Friday.
Negative reactions or quality problems may also be reported to the FDA’s MedWatch adverse event reporting program online, by phone at 800-332-1088 or by fax using this form to 800-FDA-0178.
What are nitrosamines?
Nitrosamines are present in water and certain foods such as cured and grilled meats, dairy products and vegetables. They can form in drugs during the manufacturing process, the FDA notes. Long-term ingestion of nitrosamines may be associated with an increased cancer risk in humans.
Though everyone is exposed to some level of nitrosamines, the FDA set an acceptable daily intake limit for the impurity. A person taking a drug containing the impurity at or below the acceptable daily limit every day for 70 years is not expected to have an increased cancer risk.