AARP Hearing Center
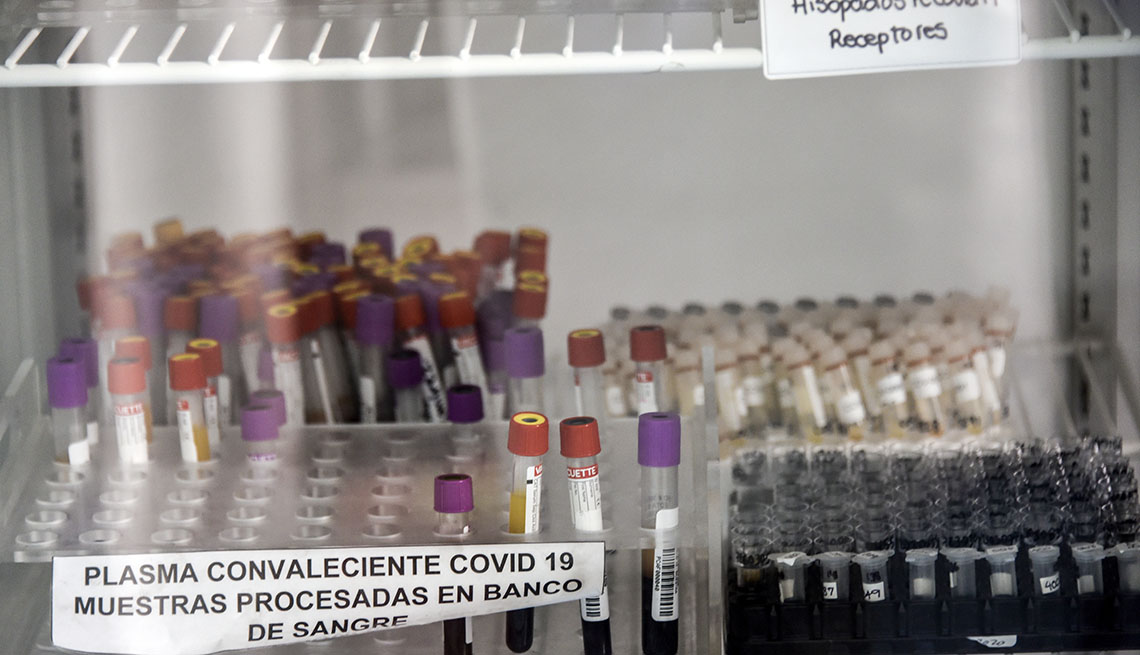
The U.S. Food and Drug Administration (FDA) announced Sunday it will now allow emergency use of “convalescent plasma therapy” for hospitalized coronavirus patients — a move that brought hope while it stirred debate.
The treatment, which involves the injection of the liquid component of blood that contains antibodies from those who have recovered from the coronavirus, is thought to be fairly safe. Early results of a study from the Mayo Clinic showed it could, under certain limited circumstances, boost the body’s immune system to fight the virus — if given early enough.
However, that study is still underway, and the data released so far does not meet the widely established standard of a double-blind, peer-reviewed investigation involving a placebo control group, as the Mayo researchers themselves readily acknowledged. Specifically, they reported that the seven-day mortality rate among 35,000 mostly critically ill patients who were transfused with the therapy within three days of being diagnosed with COVID-19 was lower (8.7 percent) than for patients transfused with the therapy four days after diagnosis (11.9 percent).
News of the treatment’s emergency approval — which reversed an earlier decision — comes as a large, nationwide, rigorous scientific trial is underway to compare the safety and efficacy of convalescent plasma therapy versus a placebo among adults exposed to COVID-19. The double-blind study is being sponsored by the National Institutes of Health in Bethesda, Maryland, and the Department of Defense.