AARP Hearing Center
A new technology will eliminate the need for fingerstick testing among many adults who need to monitor their glucose levels for diabetes treatment.
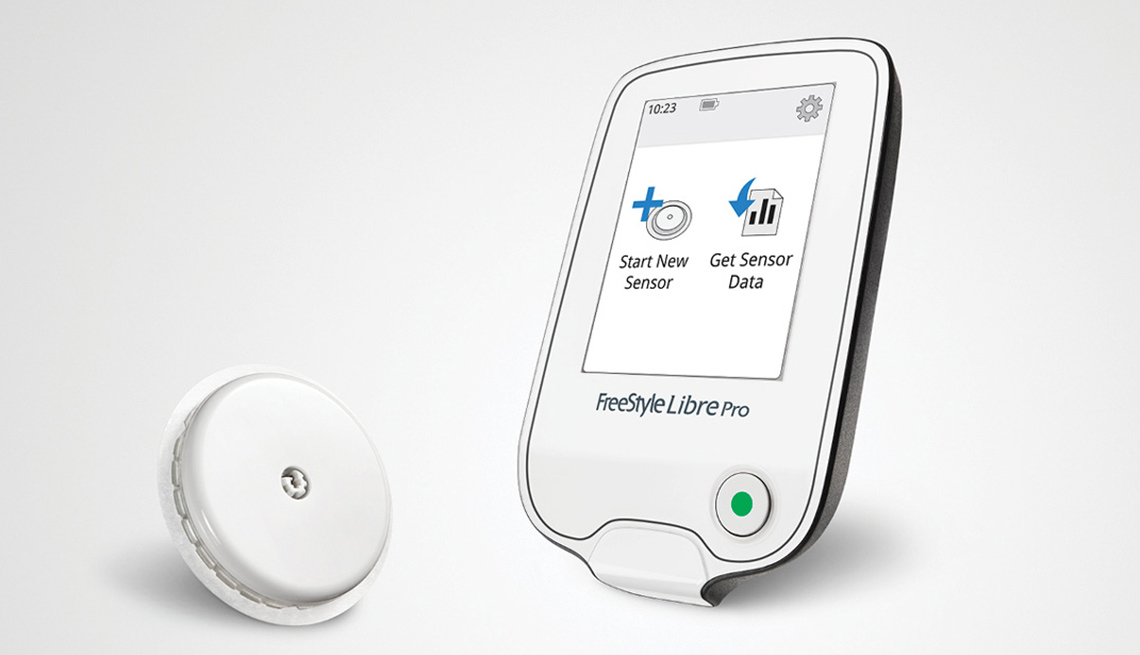
The Food and Drug Administration (FDA) this week approved the FreeStyle Libre Flash Glucose Monitoring System, which continuously monitors glucose, or sugar, levels by means of a sensor wire inserted below the skin's surface. With the device, adult patients can determine glucose levels by waving a mobile reader above the wire to determine whether levels are too high or too low, without the need to draw a blood sample from their fingertips.
It has been common practice for diabetes patients to regularly test their blood sugar levels, often many times a day, using the fingerstick method, then testing the sample with a blood glucose meter.
Donald St. Pierre, deputy director of new product evaluation for the agency's Center for Devices and Radiological Health, said the new system, manufactured by Abbott Diabetes Care Inc., allows patients “to avoid the additional step of fingerstick calibration, which can sometimes be painful ... with a wave of the mobile reader.”
The Centers for Disease Control and Prevention reports that more than 29 million people in the United States have diabetes, either because their bodies do not make enough insulin (type 1) or because they do not process it properly (type 2). Insulin is essential in controlling blood sugar, and high blood sugar levels can lead to heart disease, stroke, kidney failure and other ailments.