Heartburn Drug Recalls Continue Over Fears of Cancer Risks
Feds continue to investigate potential harm from contaminant
En español | Three drug companies are recalling popular heartburn medications that may contain “unacceptable levels” of a possible cancer-causing substance, federal officials say.
The recalls, posted by the Food and Drug Administration (FDA) this week, are the latest in a series that began in September 2019, when the FDA alerted the public to the presence of a probable human carcinogen, N-Nitrosodimethylamine (NDMA), in some ranitidine medications. Ranitidine is the generic form of Zantac. It is available over the counter and by prescription.
The FDA's announcement prompted national drugstore chains to yank ranitidine medications from their shelves “out an abundance of caution.” Several drug manufacturers followed with voluntary recalls for their medications. Now, more are joining in.
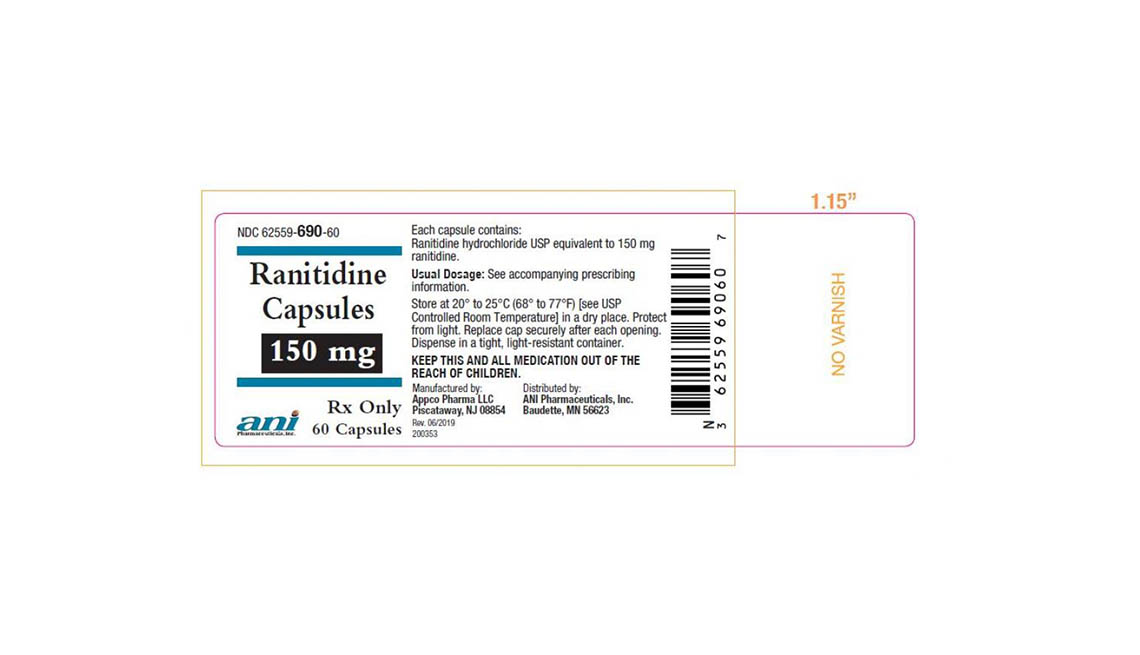
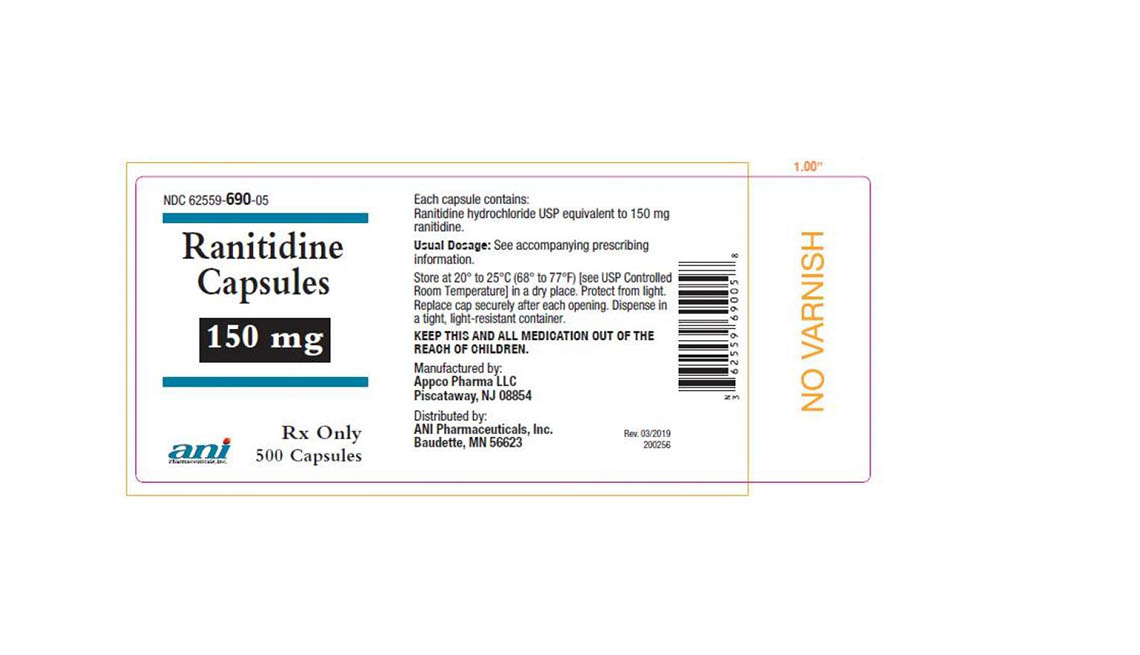
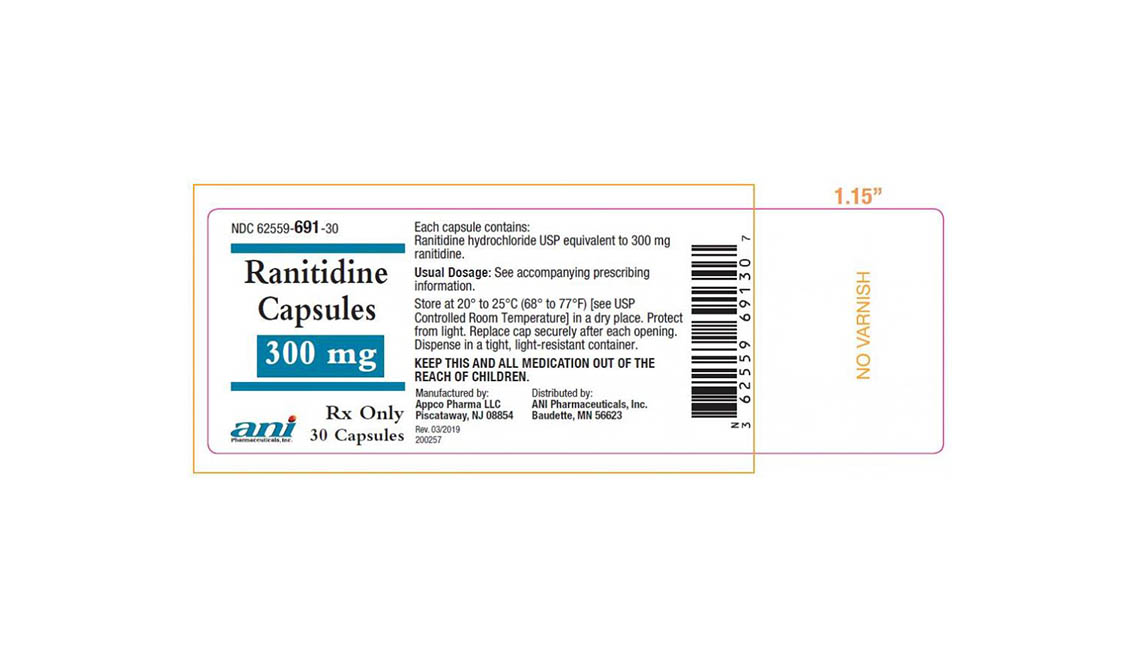
Denton Pharma Inc. is recalling all of its unexpired 150-milligram and 300-milligram ranitidine tablets; Appco Pharma is recalling all of its unexpired 150-milligram and 300-milligram ranitidine hydrochloride capsules.
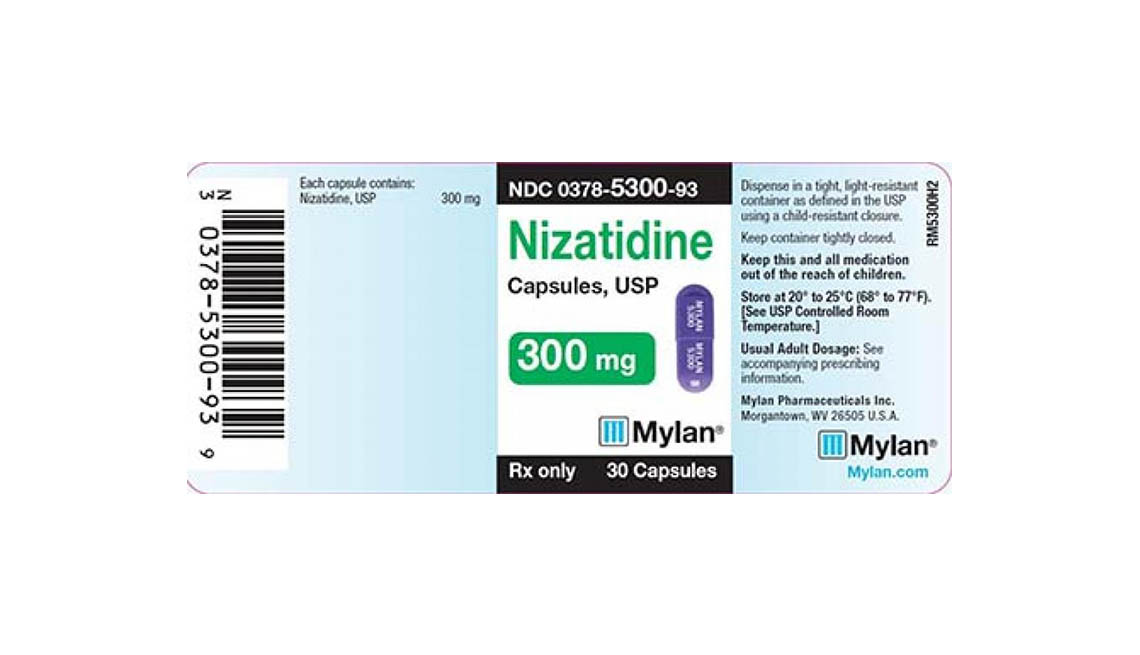
Mylan Pharmaceuticals also issued a recall this week for three batches of nizatidine capsules — an antacid used to treat ulcers and heartburn due to gastroesophageal reflux disease.
All the companies cited NDMA impurities or potential impurities in the pills as the reason for the recalls, but say no adverse health effects have been reported so far. Consumers are still being advised to stop taking the medication immediately.
NDMA is a known environmental contaminant typically found in water and foods, including meats, dairy products and vegetables, according to the FDA. Recently, the agency launched an investigation to “understand the cause of this impurity in these drugs.”
For expert tips to help feel your best, get AARP’s monthly Health newsletter.
The FDA has also been investigating NDMA impurities in other medications, including blood pressure and heart failure drugs, since 2018, and is currently “evaluating whether the low levels of NDMA in ranitidine pose a risk to patients.”
People taking prescription ranitidine who wish to stop should talk to their health care provider about other treatment options, the FDA says. Government testing has not found NDMA in other common heartburn and ulcer medications, including Pepcid (famotidine), Tagamet (cimetidine), Nexium (esomeprazole), Prevacid (lansoprazole) and Prilosec (omeprazole).
Lifestyle modifications can also help reduce the symptoms of heartburn, says Brian Antono, a family medicine physician and health policy fellow at Georgetown University School of Medicine in Washington, D.C.
In an earlier interview with AARP, he recommended limiting caffeine, avoiding spicy foods and steering clear of other triggers that cause acid reflux.
Mylan Pharmaceuticals says consumers who have its recalled products (150-milligram and 300-milligram nizatidine capsules with lot numbers 3086746, 3082876 and 3082877) should contact Stericycle at 888-628-0727 to return the medication.
Consumers with questions regarding Appco Pharma's recall should call 732-253-7735 or e-mail pv@appcopharma.com. Those who purchased Denton Pharma's impacted product can call 800-722-0772 to arrange for product return.
If you notice any problems that may be related to the recalled drugs, consult your physician, the FDA says. Adverse reactions should also be reported to the FDA's Safety Information and Adverse Event Reporting Program.
Below are product photos of the recalled medications.
- |
- Photos