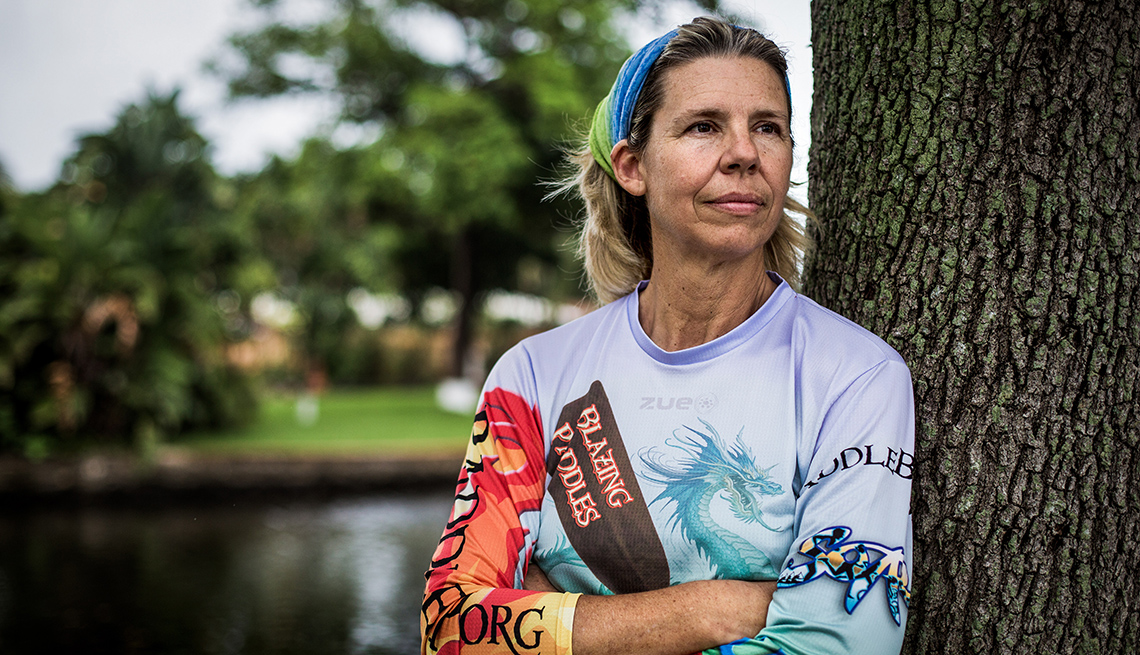Staying Fit
Over the last couple years, Judy Perkins has lived life according to her bucket list. She went rafting in the Grand Canyon, and hiking in Nepal and Patagonia. She completed a trip around Florida by sea kayak.
Perkins, 52, did all these things because she believed she would die within months of metastatic breast cancer. She was first diagnosed and treated for early-stage breast cancer — ductal carcinoma in situ — in 2003. She opted for aggressive treatment: a mastectomy, where surgeons removed her entire left breast. She assumed she was cured. Then, a decade later, she felt a lump on that same side, where her breast had once been. The cancer had recurred, but this time it had spread throughout her chest wall. She had metastatic breast cancer, and her oncologist gave her three years to live.


AARP Membership— $12 for your first year when you sign up for Automatic Renewal
Get instant access to members-only products and hundreds of discounts, a free second membership, and a subscription to AARP the Magazine.
Over the next two years, Perkins hunted for clinical trials to join and tried every treatment imaginable. She had some success with one drug but had to stop taking it after developing a dangerously low platelet count. In 2015, just as she felt she'd run out of options, she heard about a clinical trial at the National Institutes of Health using a new type of therapy, called immunotherapy, to treat cancer.
She flew to Bethesda, Md., to undergo treatment — a grueling ordeal that involved (along with five days of high-dose chemotherapy) having doctors extract her own immune cells, enhance them in a laboratory, then inject them back into her body. She experienced high fevers, chills and bouts of diarrhea. But within about a week, she realized the treatment was working. “The tumor had been pressing on a nerve that sent pain shooting all the way up and down my chest and back,” she says. “The pain disappeared so rapidly that 10 days after treatment, I was able to completely stop taking my pain medications.”
More than two years later, Perkins is cancer free, and doctors have told her they don’t expect her cancer to come back. “It’s so strange, because three years ago I was planning my bucket list and thinking about all the books I would read before I died,” she says. “Now I have a new lease on life. I have probably 20 or 30 years ahead of me. I quit my job as an engineer when I was diagnosed with metastatic cancer. Now I have to think about what I want to do — go back to nursing school? Volunteer?”
Immunotherapy — treatment that uses certain parts of a person’s immune system to fight diseases such as cancer — isn’t a new concept. “The idea that we could use the body’s own natural defense mechanisms to attack cancer cells has actually been around for quite some time, since the 1960s,” says Len Lichtenfeld, M.D., deputy chief medical officer of the American Cancer Society. But incredibly, it’s taken a half-century to come to fruition.
This revolutionary therapy has quickly become the gold standard to treat many types of hard-to-treat-cancers, such as lung cancer, melanoma and kidney cancer, says Sumanta Pal, M.D., an associate professor in the department of clinical oncology at the City of Hope Comprehensive Cancer Center.
But while immunotherapy can be a miracle therapy for some, it’s not a panacea for everyone, Pal warns. One preliminary analysis found that fewer than 10 percent of all cancer patients can benefit from it. In rare cases, it’s even been linked to harm. “There’s a rush now to try immunotherapy with end-stage cancers, but it doesn’t work for every patient and every cancer type,” Pal says. “That’s why it’s important for patients to understand the pros and cons and have an in-depth conversation with their oncologist.”
Here’s a look at what types of immunotherapy are currently available and what’s in the pipeline for the future.

Currently Available Treatments
There are two main types of immunotherapy currently approved to treat certain types of cancers: drugs called checkpoint inhibitors and the less frequently used chimeric antigen receptor (CAR) T-cell therapy. We break down both below, starting with the drugs.
1. Checkpoint inhibitors
This class of FDA-approved drugs is now being used now for many types of cancer, including melanoma, lung and kidney cancers. One drug, pembrolizumab, made headlines earlier this year when a study in the New England Journal of Medicine found that when it was combined with chemotherapy, it dramatically boosted patients' odds of surviving lung cancer.
Brand names:
- Keytruda (pembrolizumab)
- Opdivo (nivolumab)
- Tecentriq (atezolizumab)
- Bavencio (avelumab)
- Imfinzi (durvalumab)
- Yervoy (ipilimumab)